For decades, the standard of care for autoimmune diseases like multiple sclerosis (MS), lupus, and vasculitis has been defined by a strategy of management rather than resolution. Patients have relied on a lifelong regimen of immunosuppressants, hoping to dampen the inflammatory fires consuming their bodies. But for Jan Janisch-Hanzlik, a 49-year-old nurse from Nebraska, "management" was no longer enough. As her multiple sclerosis progressed—stealing her ability to work, forcing her to abandon active parenting, and anchoring her future to the looming threat of a wheelchair—she sought a radical alternative.
In June 2025, Janisch-Hanzlik became the first patient at the University of Nebraska Medical Center to undergo a pioneering procedure: CAR T cell therapy. Originally engineered as a "living drug" to hunt down and eradicate aggressive blood cancers, this therapy is now being repurposed to perform a wholesale reset of the human immune system.
The Science of Reprogramming: From Oncology to Autoimmunity
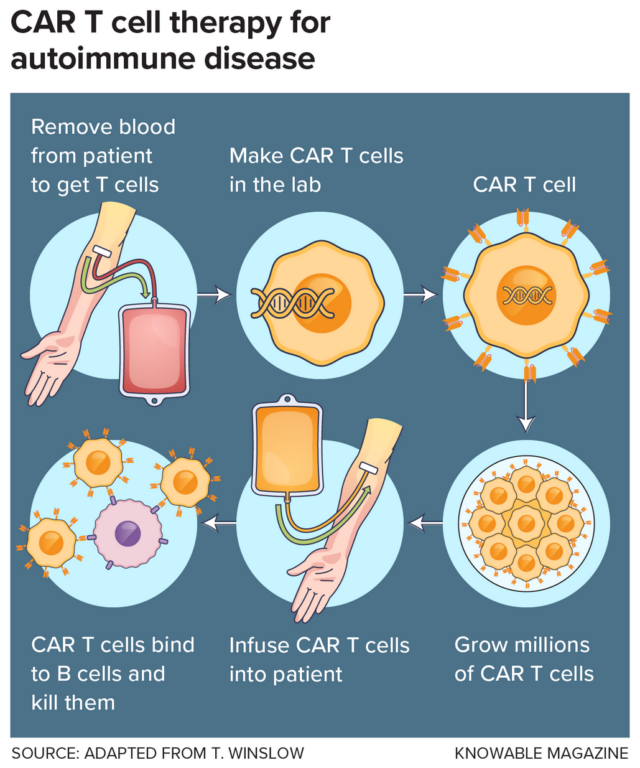
The core of CAR T (Chimeric Antigen Receptor T cell) therapy lies in the modification of the body’s own T cells. These cells are the sentinels of the immune system, typically responsible for identifying and neutralizing foreign invaders like bacteria or viruses. In cancer treatment, which received its first FDA approval in 2017, scientists extract these cells from a patient’s blood, genetically engineer them to express a receptor that recognizes a specific protein on the surface of malignant cells, and reinfuse them. Once inside, these "super-charged" cells hunt down the cancer with lethal precision.
In the context of autoimmune diseases, the target is different but the logic remains identical. Autoimmunity occurs when B cells—the immune cells responsible for producing antibodies—malfunction, misidentifying the body’s own tissues as threats. By programming T cells to recognize and eliminate these rogue B cells, clinicians believe they can effectively "wipe the slate clean," allowing the immune system to reboot without the cellular "memory" that drives the disease.
A Chronology of Discovery
The bridge between oncology and rheumatology was first built in Germany. In 2021, a research team reported a breakthrough after treating a patient with systemic lupus erythematosus (SLE) using CAR T cells. The results were unprecedented: the patient achieved long-term remission, with symptoms vanishing and the disease markers stabilizing in a way that traditional biologics had never achieved.
Following this success, the medical community accelerated its efforts to test the therapy across a broader spectrum of conditions:
- 2021–2023: Early proof-of-concept trials in Germany and the US demonstrate the viability of B-cell depletion in autoimmune patients.
- December 2025: Kyverna Therapeutics announces positive topline data from a study involving 26 patients with stiff-person syndrome. Participants showed significant improvements in mobility, with many abandoning walkers and canes entirely within 16 weeks.
- June 2025: Jan Janisch-Hanzlik receives the first "off-the-shelf" CAR T infusion at the University of Nebraska Medical Center, marking a shift toward donor-derived therapies.
- Early 2029 (Projected): Completion of major clinical trials currently assessing the long-term safety and efficacy of these interventions.
Supporting Data: Evidence of Potential
The preliminary data emerging from these trials has been described by many in the field as a "game changer." In the study led by neurologist Amanda Piquet of the University of Colorado Anschutz, 26 patients with the rare, debilitating stiff-person syndrome were treated with a single dose of CAR T therapy. The results were striking: by the four-to-12-month follow-up mark, every single patient had ceased the use of other systemic immunotherapies.
Moreover, the "reboot" does not appear to leave patients defenseless. Studies have shown that while the CAR T cells eliminate the pathogenic B cells, they often leave behind older, "memory" B cells that retain immunity to previously encountered pathogens like measles or chickenpox. This suggests the immune system remains functional, even as the specific autoreactive components are purged.
Risks, Uncertainties, and the "Sticker Shock"
Despite the optimism, the transition from cancer to autoimmunity is fraught with complexity. CAR T therapy is an intensive medical intervention that carries significant risks.
Inflammatory Complications
When CAR T cells attack their targets, they trigger a massive release of cytokines—proteins that can cause high fevers, dangerous drops in blood pressure, and, in severe cases, neurotoxicity. While clinicians have spent a decade refining the management of these side effects, they remain a significant hurdle.
The Problem of Long-Term Toxicity
In February 2026, the FDA issued a cautionary note regarding the "unpredictable long-term toxicity" of the treatment. There is legitimate concern that the genetic modifications used to create these cells could, in rare instances, lead to the development of secondary T-cell-based cancers. For a cancer patient with a poor prognosis, this risk is acceptable; for an autoimmune patient, the calculus is more difficult.
The Economic Barrier
The financial burden of CAR T therapy is currently astronomical, often reaching hundreds of thousands of dollars per patient due to the need for personalized laboratory engineering. Experts like Matt Lunning, medical director for gene and cellular therapy at Nebraska Medicine, emphasize that for this therapy to be a viable standard of care, the field must shift toward "off-the-shelf" models—using donor cells rather than patient-specific ones—or developing ways to engineer cells in vivo (within the body), which would drastically reduce costs.
Implications: A New Era of Medicine
The case of Jan Janisch-Hanzlik serves as both a testament to the power of modern medicine and a reminder of the uncertainty inherent in pioneering. Nearly a year after her infusion, Janisch-Hanzlik has reclaimed significant portions of her life. She no longer requires corrective lenses for double vision, her reliance on a cane is a thing of the past, and her daily need for restorative, multi-hour naps has vanished.
Yet, the clinical journey continues. She still navigates the residual effects of her MS—numbness in her feet, muscle weakness, and occasional word-finding difficulties. When she asks her physicians about her long-term prognosis, the answer remains the same: "We don’t know. You’re the first."
The future of CAR T in autoimmune disease hinges on the next generation of therapies, such as those using mRNA-based encoding. By using short-lived genetic messengers rather than permanent DNA alterations, researchers like James Howard at the University of North Carolina are developing therapies that "turn off" after the job is done, theoretically eliminating the risk of long-term malignancy and reducing the potential for systemic inflammation.

As the scientific community watches these trials unfold, the medical establishment is forced to confront a paradigm shift. If we can successfully "reset" the immune system rather than merely suppressing it, we may be on the cusp of curing diseases that have historically been considered life sentences. For patients like Janisch-Hanzlik, the risk is a secondary concern to the possibility of a life restored.
"I would want to be able to say I did everything that I possibly could to prevent them, or anyone else, from having something like this," Janisch-Hanzlik says, referring to her grandchildren who may carry the genetic markers for her disease. Her participation in this study is not just an act of survival; it is an investment in a future where autoimmune diseases are no longer managed, but resolved.